Atoms All matter is made of atoms. They are the building blocks of matter.
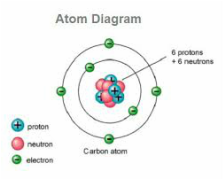 Picture of an atom's structure.
Picture of an atom's structure.
Subatomic Particles
Particles that are smaller than the atom are called Subatomic Particles. They have three parts.
The proton and the neutron are found in the center of the atom, which is the nucleus. The electrons orbit the nucleus.
Atoms always have as many electrons as protons. Atoms ususally have about as many neutrons as protons.
1. Adding a proton makes a new kind of atom. Adding a neutron makes an isotope of that atom, a heavier version of that atom.
Particles that are smaller than the atom are called Subatomic Particles. They have three parts.
- Proton: Positive
- Neutron: no charge
The proton and the neutron are found in the center of the atom, which is the nucleus. The electrons orbit the nucleus.
Atoms always have as many electrons as protons. Atoms ususally have about as many neutrons as protons.
1. Adding a proton makes a new kind of atom. Adding a neutron makes an isotope of that atom, a heavier version of that atom.

The top number, 11 in this case, is the atomic number. This is also the number of protons the element contains.
The NA is where the element symbol goes. Under this is the elements name and after is the Atomic Mass/Weight , which is the number of protons and neutrons added together.
Electons live in something called shells or energy levels distances away from the nucleus and are broken into sublevels. There are seven levels, each containing a certain amount of electrons; 2(n)^2
There are four possible sublevels: s,p,d,f
Electron Energy Level Notes
An atomic orbital is a mathematical function that describes the wave like behavior of either one electron or a pair of electrons in an atom. This function can be used to claculare the probability of finding any electron of an atom in any specific region around the atoms nucleus.
The NA is where the element symbol goes. Under this is the elements name and after is the Atomic Mass/Weight , which is the number of protons and neutrons added together.
Electons live in something called shells or energy levels distances away from the nucleus and are broken into sublevels. There are seven levels, each containing a certain amount of electrons; 2(n)^2
There are four possible sublevels: s,p,d,f
Electron Energy Level Notes
- In each energy level, electrons fill sublevels in a certain order.
- Level 1 only has s sublevel ( a spherical shape )
1. 2 electrons may fit in this sublevel. - Level 2 has to 2 sublevels: s and p
1. There are two electrons in s and there are three different p orbitals and may hold 2 electrons each- 6 total making the total of level two 8 electrons.
An atomic orbital is a mathematical function that describes the wave like behavior of either one electron or a pair of electrons in an atom. This function can be used to claculare the probability of finding any electron of an atom in any specific region around the atoms nucleus.
