Matter Anything that takes up space and has mass
There are four phases: Solid, Liquid, Gas, and Plasma.
There are two types of properties of matter
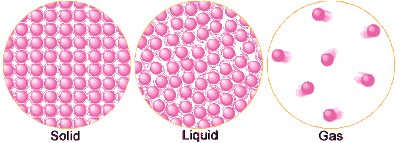
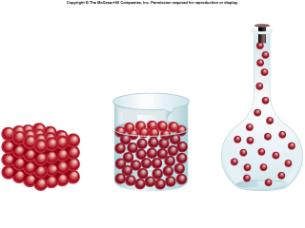
- Solids: Have definite shape and volume. Contain mass and take up space.
1. The particles are tighly packed together
2. Have little energy
3.Vibrate in place - Liquids: Have no definitive shape but do have a definitive volume.
1. Have a mass
2. Takes up space. - Gas: Have no definitive shape or volume.
1.Move freely
2.Have a lot of energy - Plasma: Similar to gas, but the particles are electricallly charged.
1. Extremely high energy levels.
There are two types of properties of matter
- Chemical: properties of matter that may only be observed and measured by performing a chemical change or chemical reaction.
Examples
- reactivity with other chemicals
- toxicity
- oxidation states
- chemical stability
- Physcial: characteristic that can be observed or measured without changing the composition of the sample.
Examples:
- Volume
- Mass
-Color
- Density
-Length
Kinetic models
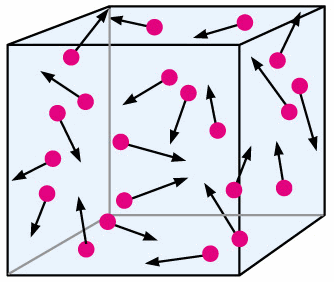
The Kinetic Theory of Matter states that all matter is made of a large number of tiny atoms or molecules which are in continous motion and has been demonsrtated by Brownian motion and diffusion.
- When randomly moving gas molecules hit the wall of a container they exert a force on the wall. As the pressure is defined as force per unit area, the force acting in the container gives rise the gas pressure.
- As temperature of a gas in container increases, the molecules move faster and hit the walls more frequently and more violently.
- Gas pressure increases with gas temperature and vice versa at constant volume.