exam
Make the following problems:
a. If an atom has 19 protons
What is the atomic number?
What is the symbol?
-Z:19
-Symbol: K (Potassium)
b. If the atomic mass is 65 and the atomic number 30
How many neutrons does the atom have?
-A: 65
-Z: 30
-Neutrons: 35
c. If an atom has 68 neutrons and the atomic mass is 118
How many electrons does the atom have? Electrons: 50
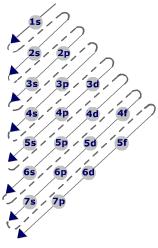
2. Draw paulings triangle, make the electronic configuration, and write: Group, period, and number of levels.
a. Hydrogen, Z:1 1s^1 Group IA, Period 1
b. Carbon, Z: 6 1s^2, 2s^2, 2p^2 Group IVA, Period 2
c. Oxygen, Z: 8 1s^2, 2s^2, 2p^4 Group VIA, Period 2
d. Sodium, Z:11 1s^2, 2s^2, 2p^6, 3s^1 Group IA, Period 3
e. Chlorine, Z:17 1s^2, 2s^2, 2p^6, 3s^2, 3p^5 Group VIIA, Period 3
3. Using Dalton's, Thompson's, Rutherford's, Bohr's Models and the modern model explain the evolution of the atom model.
It first started off with the greeks stating that all matter is made of four elements: fire, water, earth, and air. After the greeks came Cassandi and Boil testing more experiments. John Dalton made a model of an atom as a solid and an invisible sphere. stated the theory that all matter is made of atoms and stated the theory of compouds made of atoms of different elements. After John Dalton comes Jospeh Thompson who discovered negative particles, or electrons. This led to the change of the atomic model. Ernest Rutherford made the gold foil experiment, which resulted in alpha particles that were positive. This resulted in the discovery of the protons. Bohr discovered that atoms had energy levels, or shells, that surrounded the nucleus with electrons orbiting around them.
Weiner Heisenberg demonstrated that an electrons exact position can be found using a mathematical equation that follows, 2(n)^2. Lastly James Chadwick discovered the nucleus.
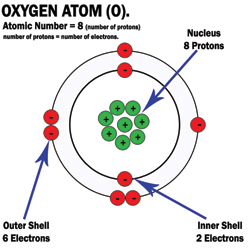
4. Draw the structure of the Oxygen atom icluding: Nucleus, orbits, protons, neutrons and electrons.
5. Define: Atomic mass, atomic number, Isotopes, level, sublevel.
- Atomic Mass: The sum of protons and neutrons in an atom
- Atomic Number: The number of protons in an atom
- Isotopes: Atoms of the same element that differ from each other's mass number.
- Level: Surround the nucleus in an atom. There are 7 maximum levels. Each level has a maximum capacity of electrons and the formula to find the electrons exact position is 2(n^2), n being the number of the level
- Sublevel: Levels are divided into sublevels which are s, p, d, and f. Each sublevel has a maximum number of electrons it can hold: s is 2, p is 6, d is 10, and f is 14.
6. How is the electronic table organized?
a. If an atom has 19 protons
What is the atomic number?
What is the symbol?
-Z:19
-Symbol: K (Potassium)
b. If the atomic mass is 65 and the atomic number 30
How many neutrons does the atom have?
-A: 65
-Z: 30
-Neutrons: 35
c. If an atom has 68 neutrons and the atomic mass is 118
How many electrons does the atom have? Electrons: 50
2. Draw paulings triangle, make the electronic configuration, and write: Group, period, and number of levels.
a. Hydrogen, Z:1 1s^1 Group IA, Period 1
b. Carbon, Z: 6 1s^2, 2s^2, 2p^2 Group IVA, Period 2
c. Oxygen, Z: 8 1s^2, 2s^2, 2p^4 Group VIA, Period 2
d. Sodium, Z:11 1s^2, 2s^2, 2p^6, 3s^1 Group IA, Period 3
e. Chlorine, Z:17 1s^2, 2s^2, 2p^6, 3s^2, 3p^5 Group VIIA, Period 3
3. Using Dalton's, Thompson's, Rutherford's, Bohr's Models and the modern model explain the evolution of the atom model.
It first started off with the greeks stating that all matter is made of four elements: fire, water, earth, and air. After the greeks came Cassandi and Boil testing more experiments. John Dalton made a model of an atom as a solid and an invisible sphere. stated the theory that all matter is made of atoms and stated the theory of compouds made of atoms of different elements. After John Dalton comes Jospeh Thompson who discovered negative particles, or electrons. This led to the change of the atomic model. Ernest Rutherford made the gold foil experiment, which resulted in alpha particles that were positive. This resulted in the discovery of the protons. Bohr discovered that atoms had energy levels, or shells, that surrounded the nucleus with electrons orbiting around them.
Weiner Heisenberg demonstrated that an electrons exact position can be found using a mathematical equation that follows, 2(n)^2. Lastly James Chadwick discovered the nucleus.
4. Draw the structure of the Oxygen atom icluding: Nucleus, orbits, protons, neutrons and electrons.
5. Define: Atomic mass, atomic number, Isotopes, level, sublevel.
- Atomic Mass: The sum of protons and neutrons in an atom
- Atomic Number: The number of protons in an atom
- Isotopes: Atoms of the same element that differ from each other's mass number.
- Level: Surround the nucleus in an atom. There are 7 maximum levels. Each level has a maximum capacity of electrons and the formula to find the electrons exact position is 2(n^2), n being the number of the level
- Sublevel: Levels are divided into sublevels which are s, p, d, and f. Each sublevel has a maximum number of electrons it can hold: s is 2, p is 6, d is 10, and f is 14.
6. How is the electronic table organized?